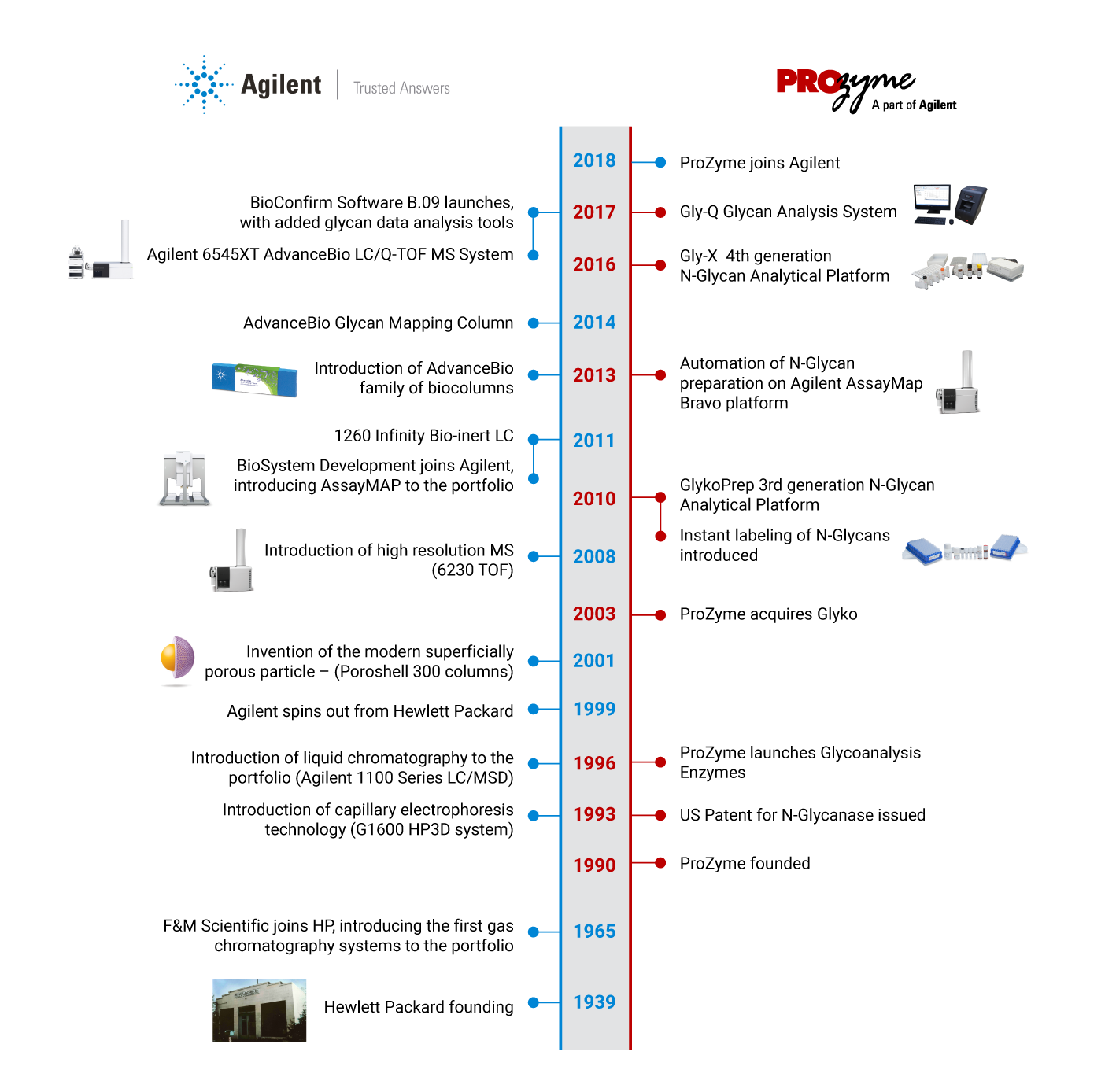
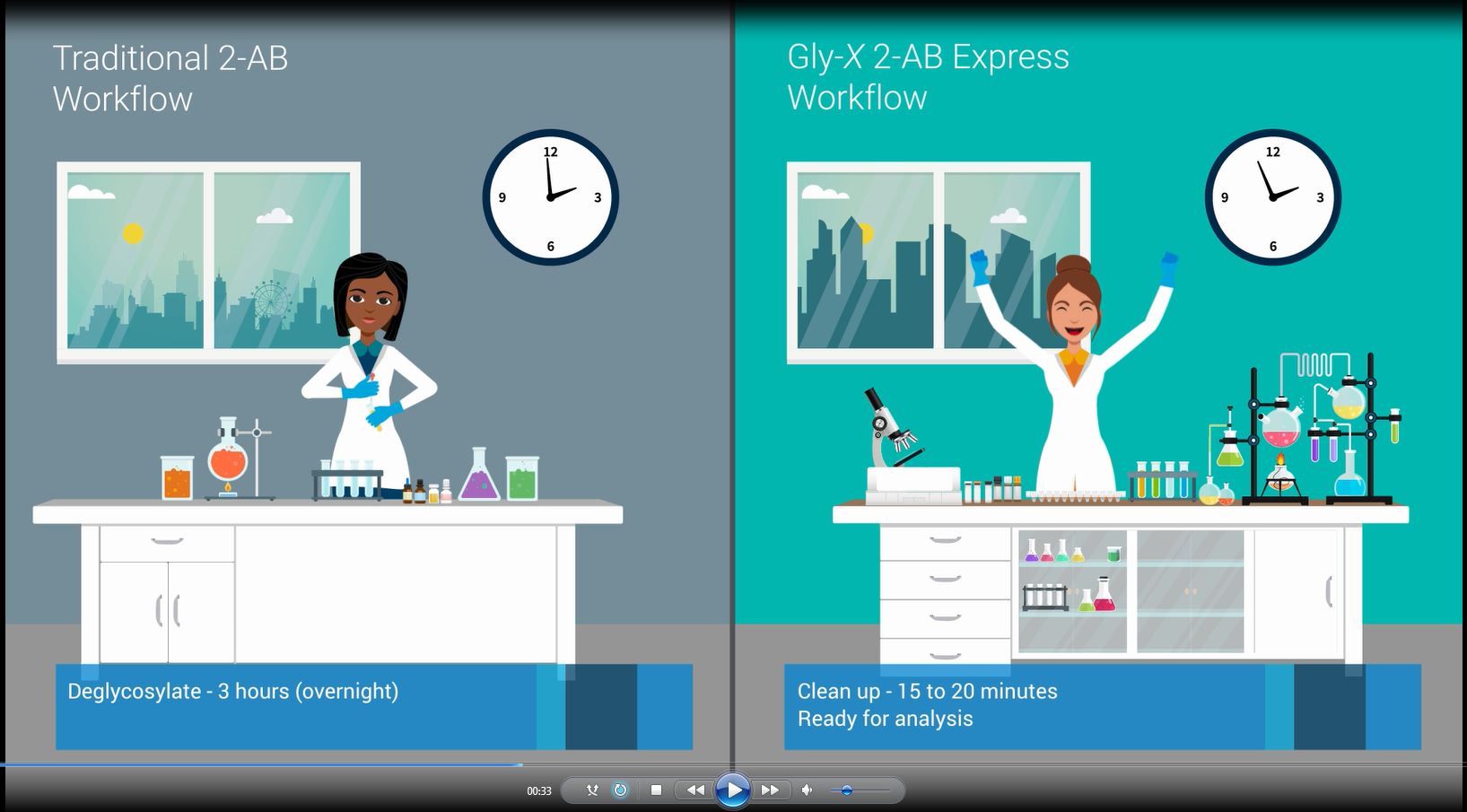
With the addition of ProZyme products and services, Agilent provides a single source offering for instruments and consumables, from sample to trusted answer. Our expertise now covers the complete glycan analysis workflow,
in addition to other critical quality attributes, so you can easily get the reliable, reproducible results you need – fast.
We are better together.
Browse our new glycan analysis solutions and bioanalytical proteins:
If you would like to request a demo, speak to an Agilent representative, or wish to be kept up-to-date on latest news and offers, complete the form below.